|
Through the electrodes, an electricity can be applied to create an electric current through the center of the tube. 
The tube has two electrodes or metal pieces on each side of the tube. A vacuum is created by removing all the air, which also removes all the matter in the space. In 1913 Thomson published an influential monograph urging chemists to use the mass spectrograph in their analyses. A cathode-ray tube is a glass tube with a vacuum space inside. His nonmathematical atomic theory-unlike early quantum theory-could also be used to account for chemical bonding and molecular structure (see Gilbert Newton Lewis and Irving Langmuir). Of all the physicists associated with determining the structure of the atom, Thomson remained most closely aligned to the chemical community. He was a good lecturer, encouraged his students, and devoted considerable attention to the wider problems of science teaching at university and secondary levels. Thomson, led to the discovery of the negatively charged part of the atom, the electron. Even though he was clumsy with his hands, he had a genius for designing apparatus and diagnosing its problems. The meaning of CATHODE-RAY TUBE is a vacuum tube in which a beam of electrons is projected on a phosphor-coated screen to produce a luminous spot at a point. Thomson announced the results of his previous four months experiments on cathode rays. The cathode ray tube experiment, originally carried out by J.J. How Cathode Rays Works The electrode at the negative end is called a cathode. In 1884 he was named to the prestigious Cavendish Professorship of Experimental Physics at Cambridge, although he had personally done very little experimental work. A cathode ray is a beam of electrons in a vacuum tube traveling from the negatively charged electrode (cathode) at one end to the positively charged electrode ( anode) at the other, across a voltage difference between the electrodes. He was then recommended to Trinity College, Cambridge, where he became a mathematical physicist. This module takes readers through experiments with cathode ray tubes. Instead young Thomson attended Owens College, Manchester, which had an excellent science faculty. The 19th and early 20th centuries saw great advances in our understanding of the atom. 
His father intended him to be an engineer, which in those days required an apprenticeship, but his family could not raise the necessary fee. The cathode ray oscilloscope is a very rapid X-Y plotter that can show a signal which is input versus another signal or time. 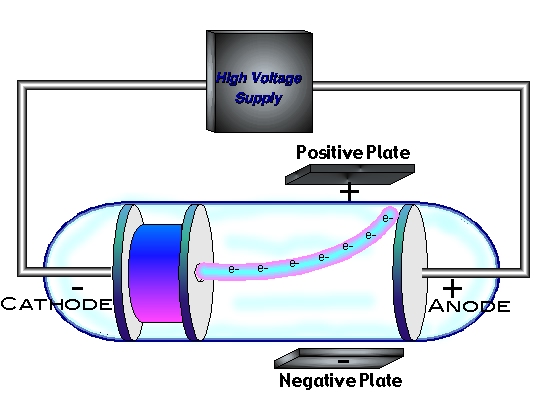
Ironically, Thomson-great scientist and physics mentor-became a physicist by default. A cathode ray oscilloscope is an appliance which has general applications in a laboratory to exhibit or to calculate and examine the various types of waves of electrical circuit. From "The Growth of Physical Science," by Sir James Hopwood Jeans (Cambridge: Cambridge University Press, 1948) Early Life and Education
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
